




1-methylhexyl, stable in neutral and acidic environments, suitable for hydrolysis under alkaline conditions.
Common name: cloquintocet-mexyl (detoxicated quine/detoxicated quine)
Chemical name: 1-methylhexyl (5-chloroquinolin-8-yloxy)acetate
Structure name: 1-methylhexyl [(5-chloro-8-quinolinyl)oxy]acetate CAS RN [99607–70–2] Development codes CGA 185072 (Ciba-Geigy) Smiles code
1-methylhexyl is an herbicide antidote developed by Novartis (now Syngenta).
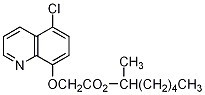
Chloroquine chemical name (5-chloroquinolin-8-methoxy) acetic acid (1-methylhexane).
The pure product is a colorless solid with a melting point of 69.4°C(61.4~69.0%C) and a vapor pressure of 5.31x10-3mPa(20°C). Allocation coefficient KowlogP=5.03(25°C). Henry constant 3.02xlO-1Pam3mo-1 Relative density 1.05. Solubility (25℃, g/L): 0.59mg/L in water, ethanol 190, acetone 340, toluene 360, n-hexane 0.14. Stable to acid, hydrolyzed to base.
Toxic rats and mice acute oral LD50gt; 2000mg/kg, acute percutaneous LD50gt; 2000mg/kg, LC50(4h):>0.935mg/L air. No irritation to skin and eyes. NOEL data: The rat (2a) dose was 44mg/kg·d. ADI value :0.04mg/kg·d, LD50gt for partridge and wild duck at acute mouth; 2000mg/kg, fish poison LC50(96h,mg/L): rainbow trout and carp gt; 76, big sunfish gt; Fifty-one. Bee LD50(48h)gt; 100μg/ piece (oral and contact). LD50> 1000mg/kg soil.
Method of using chloroquine
It is a safety agent of clodinafop-propargyl. Chloroquine is mixed with acetylidene (1:4) for weeding cereal crops.
Package
